Public
Aptar CSP News
Aptar CSP Technologies Documents
Aptar CSP Technologies Locations
Aptar CSP Technologies Videos
Subsidiaries
Maxwell Chase Technologies
If this is your company, CONTACT US to activate Packbase™ software to build your portal.


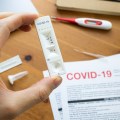
AptarGroup, Inc., a global leader in drug delivery, consumer product dispensing and active packaging solutions, announced that its Activ-Film technology was selected to protect a new SARS Rapid Antigen test for COVID-19 that recently received Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration (FDA).
The QuickVue SARS Antigen test is a point-of-care rapid antigen test developed by Quidel Corporation, a leading manufacturer of diagnostic healthcare solutions, that delivers test results in 10 minutes. The visually read test requires no supplemental instrumentation and offers expanded access to affordable and accurate COVID-19 testing that will help meet the urgent testing needs of the global economy, including for those in school systems and rural areas.
Aptar CSP Technologies’ Activ-Film technology is integrated into the diagnostic kit to protect against moisture and other environmental conditions that could otherwise impact test accuracy. Activ-Film leverages Aptar’s proprietary 3-Phase Activ-Polymer technology, which provides a broad spectrum of custom-engineered protection in a variety of configurations, such as Activ-Vial for housing diagnostics dipsticks and Activ-Tab integrated within diagnostic cassettes. This material science-based active packaging technology is currently used to protect a range of electrochemical, lateral flow and molecular diagnostic test kits on the market today.
“We are pleased to partner with Quidel® Corporation on this critical diagnostic tool and help bring the QuickVue® SARS Rapid Antigen test to the market,” said Stephan B. Tanda, Aptar President and CEO. “Our material science Activ-Film™ technology protects the test strips, helping to provide quick, reliable results to patients, their families and healthcare workers. We will continue to live up to our purpose and responsibility to society by providing solutions that protect critical COVID-19 diagnostic kits, along with dispensing solutions for medicines and consumer products needed by millions of people each day.”
Badre Hammond, Vice President Commercial Operations, Aptar CSP Technologies concluded, “As we continue to navigate through the COVID-19 crisis, this game-changing solution will help meet the urgent demand for COVID-19 testing in communities around the world. We are committed to leveraging our material science expertise to enable our partners to meet the ongoing need for innovative healthcare solutions that help improve and save lives.”




.jpg)
.jpg)




.jpg)
.jpg)













.jpg)



.png)






