Public
NEOPAC The Tube 3D Catalog
NEOPAC The Tube Competences
NEOPAC The Tube EcoDesign Collection
NEOPAC The Tube Locations
NEOPAC The Tube News
NEOPAC The Tube Videos
Subsidiaries
3D Neopac
If this is your company, CONTACT US to activate Packbase™ software to build your portal.
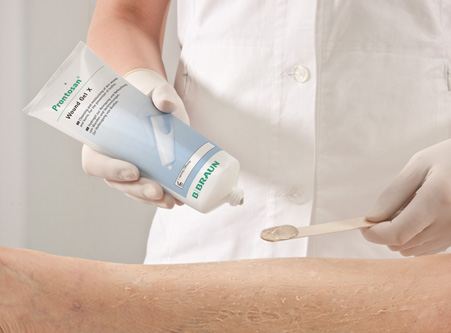

As large containers (>200 g) are required for burn surgery, B. Braun decided to extend the range of commercially available Prontosan® products with a sterile, highly viscous gel. Prontosan® Wound Gel, which was initially available only in 30-g plastic ampoules, can now be purchased in 250-g tubes. This allows straightforward, extensive application of the product to wounds.
In addition to excellent barrier properties, a new form of heat-sterilizable packaging was required. As Neopac produces its pharmaceutical tubes in fully certified clean rooms and monitors for particles and microbes, B. Braun chose Neopac as a partner for this project and got the latter well and truly on board.
Together with Neopac, the B. Braun team worked to develop an optimal packaging solution. After a few tests had been carried out on various tube materials, the results showed that a polypropylene Polyfoil® Tube was the ideal packaging for this product since this material can withstand exposure to an extreme temperature of 125° Celsius.
Polyfoil tubes from Neopac also hold their shape very well. Consequently, less air is drawn through on removing the product and the risk of contamination is reduced. Thanks to the excellent barrier properties, the tubes are extremely impermeable to organic and chemical substances and protect formulations against excess moisture or oxygen.
“The requirements for a class III medical device are stringently regulated by the Swiss decree governing Medical Devices”, explained Mr. Ruedi Hofstetter, Head of Packaging Technology at B. Braun Medical AG. “Neopac not only supported us in drafting the registration documentation but also met our flexible delivery dates”.
B. Braun Medical AG, Switzerland, is a subsidiary of the German company, B. Braun, one of the world’s leading manufacturers and suppliers of medical devices. The company employs over 44,000 people in over 50 countries.
B. Braun Medical AG has four sites in Switzerland: Sempach, Escholzmatt, Crissier and Lucerne. The Head Office of B. Braun Medical AG, Switzerland is located in Sempach. It interlinks and manages all of the companies operating in Switzerland under the B. Braun Holding AG umbrella.
Prontosan® Wound Gel X (class III medical device) in 250-g tubes is sold to end clients (clinics, practices, chemists and home care sectors such as Spitex) via B. Braun Direct Sales or specialised medical retailers.
The product is labelled in 24 languages and can thus be sold on the international market. The target groups are: doctors, wound specialists and nursing staff. The product is used in the management of acute and chronic wounds as well as burns.














.jpg)